Alzheimer’s
Alzheimer's disease is a progressive neurodegenerative disorder where early detection is crucial for effective intervention. Recent research indicates that retinal changes observable through OCT scans may correlate with neurodegenerative processes, offering a non-invasive avenue for early diagnosis. Our objective was to develop a system using Deep Learning/Vision Transformers capable of analyzing OCT scans to detect Alzheimer's disease by identifying specific retinal biomarkers.
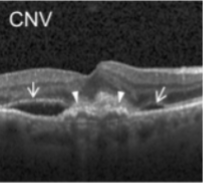
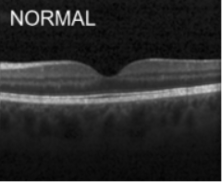
The description of each biomarker is given below:
CNV: choroidal neovascularization (CNV) with neovascular membrane (white arrowheads) and associated subretinal fluid
DME: Diabetic macular edema (DME) with retinal-thickening-associated intraretinal fluid
DRUSEN: Multiple drusen present in early AMD.
Normal: Normal retina with preserved foveal contour and absence of any retinal fluid/edema.
We compiled an approximately 84,000 OCT images dataset, categorized into four classes: CNV (Choroidal Neovascularization), DRUSEN, DME (Diabetic Macular Edema), and Normal. Extensive preprocessing was performed to enhance model performance, including:
Data Augmentation: Applied techniques like rotation, flipping, and scaling to increase data diversity.
Normalization: Standardized image pixel values to improve model training.
An EfficientNetB6 convolutional neural network was employed for model development due to its balance of computational efficiency and high performance. This architecture proved superior to previous models like ResNet50 in handling complex image classification tasks. After training, the model achieved a 97% accuracy on the test dataset, outperforming the ResNet50 model used in prior studies. This demonstrates the model's effectiveness in detecting retinal changes potentially associated with Alzheimer's disease.
Future work includes implementing optic nerve segmentation to analyze structural changes in the optic nerve and integrating other medical scans to enrich data and improve model robustness. Additionally, we plan to monitor changes in the ganglion cell layer and optic nerve head alterations observable in OCT scans.
In conclusion, our study highlights the potential of advanced deep learning models, specifically EfficientNetB6, in enhancing the detection of Alzheimer's disease through OCT scans. The high accuracy achieved signifies a promising step toward non-invasive, early diagnosis methods. Future developments could significantly improve patient outcomes by facilitating timely interventions.